DROSOPHILA IMMUNE RESPONSE
TO ENTOMOPATHOGENIC NEMATODES AND THEIR MUTUALISTIC BACTERIA
TO ENTOMOPATHOGENIC NEMATODES AND THEIR MUTUALISTIC BACTERIA
|
Due to its lack of adaptive immune response to microbial infection and availability of genetic tools, Drosophila is particularly well-suited to study innate immunity. Its history as model organism for innate immunity dated back to 1972 when Hans Boman and colleagues observed an inducible anti-bacterial immune response in D. melanogaster. |
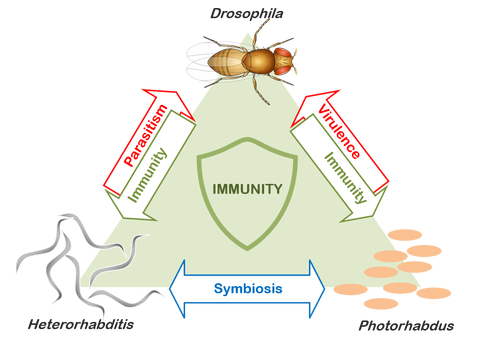
Microorganisms can directly support or compromise the development, immunity and nutrition of plants and animals, resulting in beneficial (symbiotic) or detrimental (pathogenic) outcomes for the host. Over the past decade our appreciation of the complexity of the host-pathogen dynamic has increased exponentially as we started to better clarify not only the interplay between microbes with which animals interact but also the host reactions of innate immune systems that control them. Nonetheless, despite unprecedented advances, our understanding remains incomplete and prevailing models of host-pathogen interactions fail to accommodate some key aspects evident in the relationship between invertebrates and microbes. For example, particularly understudied are the complex associations between nematodes and their symbionts as well as the molecular mechanisms of the insect immune response to the combined insult of the worms and their symbiotic bacterial pathogens.
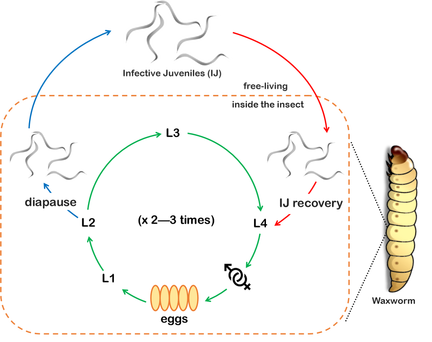
Our lab uses a tripartite system consisting of three model organisms: an insect, Drosophila melanogaster; the entomopathogenic (or insect pathogenic) nematode Heterorhabditis bacteriophora; and its symbiotic bacterium Photorhabdus luminescens, in order to investigate the molecular and evolutionary basis of insect immunity, bacterial symbiosis/pathogenicity and nematode parasitism, and to understand the basic principles of the complex interactions between these important biological processes. This system promises to reveal not only how pathogens evolve virulence but also how two pathogens can come together to exploit a common host. |
|
ROLE OF ENDOSYMBIOTIC MICROBES
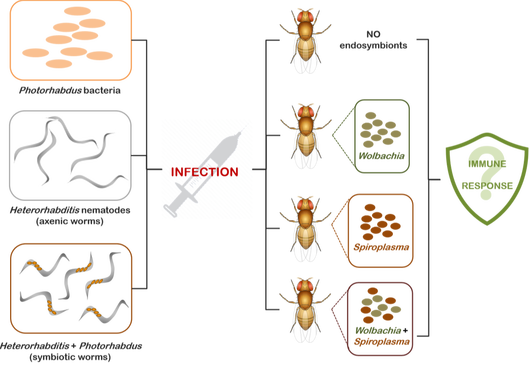
IN THE IMMUNE RESPONSE OF DROSOPHILA AGAINST NEMATODE PARASITES AND THEIR ASSOCIATED BACTERIA We investigate immune responses against Heterorhabditis nematodes and mutualistic Photorhabdus bacteria in Drosophila strains carrying or lacking Wolbachia and/or Spiroplasma endosymbiotic bacteria
Symbiotic interactions between microbes and animals are common in nature. Symbiotic microbes are particularly common in insects. In many cases, the microbial partner (usually a bacterium) lives within cells in the host’s body (it is then said to be an endosymbiont), is maternally transmitted from one host generation to another, and is never found in the free-living condition. Although such relationships have been most commonly studied with respect to nutritional effects on the host, endosymbiotic bacteria may protect their hosts from pathogenic infections. The most widespread and widely studied endosymbionts are Wolbachia and Spiroplasma, which are harbored by most insect species and are able to manipulate the reproductive properties of their hosts.
Certain Drosophila strains naturally contain Wolbachia and Spiroplasma endosymbionts, therefore the fruit fly is also considered an excellent model for investigating if/how these bacteria are able to enhance or reduce host defense mechanisms against various pathogens. Our goal is to identify the molecular basis of any changes that occur in the immune response of flies harboring endosymbiotic bacteria in the presence and absence of pathogenic infection. |
|
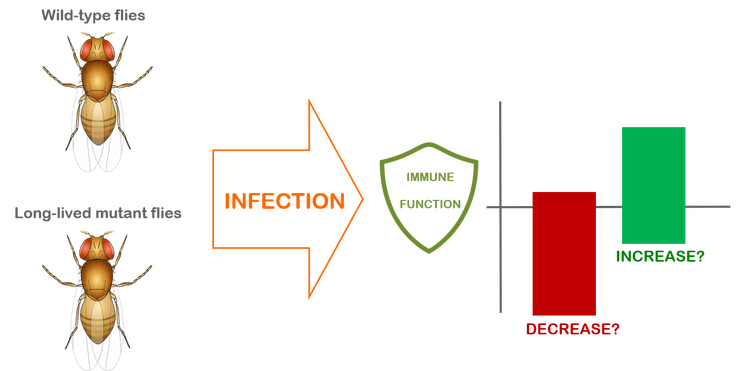
INTERACTION BETWEEN AGING AND IMMUNE MECHANISMS IN DROSOPHILA
We investigate changes in the immune function of long-lived Drosophila mutant flies compared to their wild-type controls
Aging is a complex process that involves the accumulation of deleterious changes resulting in overall decline in several vital functions, leading to the progressive deterioration in physiological condition of the organism and eventually causing disease and death. The immune system is the most important host-defense mechanism in humans and is also highly conserved in insects. Extensive research in vertebrates has concluded that aging of the immune function results in increased susceptibility to infectious disease and chronic inflammation. Over the years, interest has grown in studying the molecular interaction between aging and the immune response to pathogenic infections.
Research efforts in the areas of aging and immunity have been expanding exponentially in recent years because both fields have been favored from progress in molecular biology techniques as well as in many other research areas. The fruit fly Drosophila melanogaster is an excellent model system for dissecting the genetic and genomic basis of important biological processes, such as aging and the innate immune system, and deciphering parallel mechanisms in vertebrate animals. The overall objective of this research is to fill existing gaps in our knowledge about how aging and immunity functions are co-regulated in infected and uninfected organisms. |
DROSOPHILA ANTIVIRAL IMMUNITY AND VIRAL PATHOPHYSIOLOGY
|
Zika virus and host pathologies in Drosophila
Despite recent advances in the prevalence and epidemiological characteristics of Zika virus (ZIKV), our understanding of the molecules that participate in the host immune response to ZIKV infection, the pathophysiological defects imposed by ZIKV on the host and the specific viral components that take part in this process remain incomplete.
Using the model organism Drosophila melanogaster, our goal is to dissect host innate immune interactions with ZIKV. We investigate the mechanistic role of host factors with potential anti-ZIKV immune properties and dissect the molecular basis of the physiological activities that take place in the host during ZIKV infection. We expect that the results will generate novel insights into the immune role of host mechanisms directed against ZIKV and may expose a currently unknown layer of the innate antiviral immune system. |
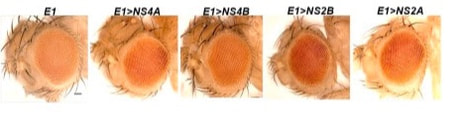
Images of the fly adult eye upon overexpression of Zika virus non-structural protein-coding genes driven under eye-specific eyeless-Gal4 (E1-Gal4)
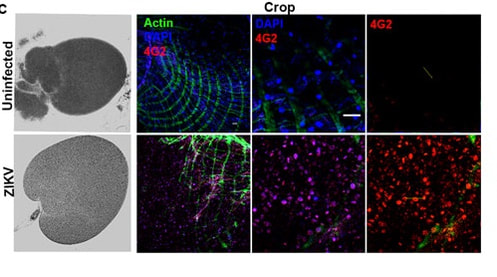
Expression of 4G2 in the crop (organ in the digestive tract) in uninfected controls and ZIKV-infected adult flies
Sources:
https://pubmed.ncbi.nlm.nih.gov/33329613/ https://pubmed.ncbi.nlm.nih.gov/32152180/ https://pubmed.ncbi.nlm.nih.gov/30305326/ |
INTERACTION BETWEEN TUMORIGENESIS WITH LIPOLYSIS AND LIPOGENESIS IN DROSOPHILA
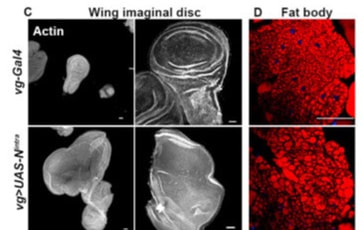
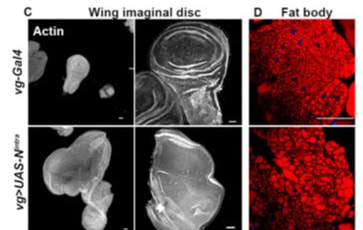
Tumors in eye and wing imaginal epithelial result in modulation of fat body lipid droplets in Drosophila larvae
Our work indicates a crosstalk between the tumor carrying tissue and the organism at the systemic level. We have found that tumor induction in imaginal epithelia triggers modulation in the fat body lipid droplets. Wing disc hyperplasia triggered by overexpression of Notch and eye disc neoplasia induced by co-expression of Notch and Mef2 leads to modulation in lipid metabolism marked by increased size of lipid droplets in the fat body. The modulation in lipid droplets is linked to altered activity of lipolysis and lipogenesis in the tumor carrying larval carcass.
These findings in combination with future studies focusing on deciphering cancer-lipid interaction will provide a platform for interpreting the role of lipid metabolism in tumor-bearing hosts, which could lead to the development of novel targeted therapeutic strategies for treating cancer.
Source: https://pubmed.ncbi.nlm.nih.gov/32946989/
These findings in combination with future studies focusing on deciphering cancer-lipid interaction will provide a platform for interpreting the role of lipid metabolism in tumor-bearing hosts, which could lead to the development of novel targeted therapeutic strategies for treating cancer.
Source: https://pubmed.ncbi.nlm.nih.gov/32946989/